providing high-precision quality and service.
Through more than 30 years of pharmaceutical logistics operations, the Sakata Group has cultivated a wide range of expertise. In addition to compliance with the PMD Act (Pharmaceutical and Medical Device Act), we provide high-precision logistics services with confidence. This includes operational management using FIFO (First-In, First-Out) by manufacturing lot, manufacturing lot/serial number history management, transport and delivery management, and the construction of logistics information systems that achieve both labor savings and high quality.
Our staff, who are well-versed in pharmaceutical logistics, act as an agent for our customers, providing support for logistics innovation and undertaking business operations while effectively utilizing our customers’ assets.
N.Osaka.Minoh Warehouse
MOVIE
(Cosmetics.3PL Logistics)
As part of our 100th-anniversary project, the Sakata Group began operations at the North Osaka Office.Minoh Warehouse in November 2019. We introduce the cosmetics logistics and 3PL services (video) at this state-of-the-art logistics center, the North Osaka Office.Minoh Warehouse.
*A web browser supporting MP4 format is required for video playback.
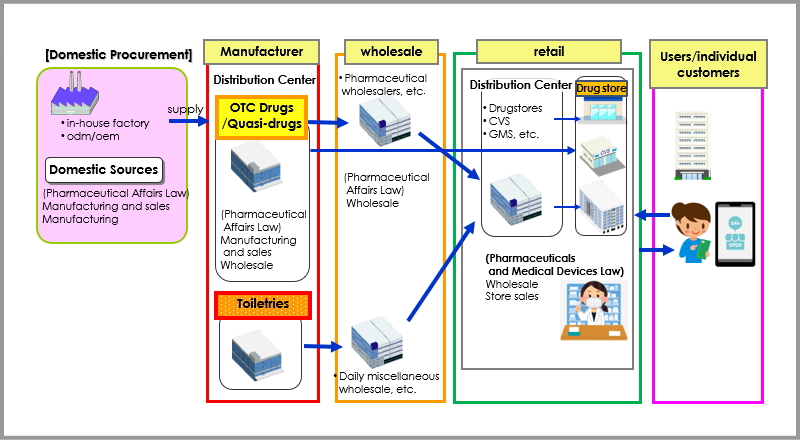
Distribution Overview of the Pharmaceutical Industry
Industry Structure (OTC Medicines + Quasi-drugs.Toiletries)
The PMD Act (Pharmaceutical and Medical Device Act) covers five areas: pharmaceuticals, quasi-drugs, cosmetics, medical devices, and regenerative medicine products. Below is a simplified diagram of the distribution channels for the pharmaceutical sector, specifically OTC medicines. Structural changes in distribution are expected to continue due to the realignment of manufacturers and wholesalers, the rise of organized retailers like drugstores, and the influence of the PMD Act.
 Value.Strengths
Value.Strengths

① Logistics Issues (Examples)
- Management by serial number is required in addition to lot management, but strict compliance has not been achieved. Furthermore, extracting history records requires significant man-hours.
- Some wholesalers return large volumes of goods, placing a heavy burden on inspection and sorting tasks.
- Due to increased inventory, there is a desire to outsource storage, but this is difficult because a wholesale general sales license and a pharmacist are required at the external warehouse.
② Customer Request Examples (Examples)
- Desire to further improve logistics services to customers and address Corporate Social Responsibility (CSR).
- Need to implement a system capable of serial number management (receiving.inventory.shipping management). Also, desire to automate and standardize operations to eliminate reliance on experienced staff.
- Desire to simplify return logistics and implement a mechanism tailored to handle returns.
- Need to find a logistics provider that can independently provide manufacturing licenses (Packaging.Labeling.Storage), wholesale general sales licenses, and pharmacists. Or find a property where such licenses can be applied for.

Sakata Warehouse 3PL Services solve these challenges

① Implementation of Serial Number Management Systems
We have implemented a management system that enables serial number management in addition to lot management. This system allows for integrated management of which products (lot.serial number) were delivered via which route from receiving to shipping.delivery, making it easy to view past records.

② Implementation of Return Inspection Systems
By introducing our independently developed return inspection system, any operator can engage in return inspection tasks in a relatively short time. By simply scanning product barcodes and manually entering lot numbers, the system cross-references returned products against past shipping data and provides voice responses for good/defective product determination.

③ Proposals for Licensed Pharmaceutical Warehouses
By appointing our own pharmacists, we can obtain pharmaceutical manufacturing licenses and wholesale general sales licenses. It is also possible to obtain a pharmaceutical manufacturing license under the customer’s name at our warehouse. We address various needs, such as setting up inspection rooms and designating stock areas for products awaiting shipping approval.

④ Inventory Management System Implementation/Integration
Data integration is possible between our proprietary inventory management system and existing ordering systems. In addition to basic inventory management, the system can provide replenishment instructions for product shortages against orders and manage the lending of fixtures and equipment.

⑤ Constant-Temperature Warehouse Proposals
By installing constant-temperature storage within our warehouses, we can propose storage and inventory management functions tailored to various product characteristics required for pharmaceutical logistics.

① Enhanced Traceability and Clearer Accountability
Previously, investigating product origins required significant man-hours to trace shipping label copies and serial number records. By implementing a history search system, administrative man-hours were significantly reduced. Furthermore, in the event of a defect, products can now be traced back to the production line and sequence, clarifying the cause and accountability among factories or suppliers.

② Reduced Workload and Improved Capacity for Returns
By implementing a system based on years of return inspection operations, workers can become effective contributors in a short time regardless of experience. This also leads to an overall increase in processing speed.

③ Enabling Outsourcing of Logistics Operations
Previously, much of the logistics had to be managed in-house due to manufacturing license constraints. However, outsourcing is now possible as we provide warehouses with the necessary licenses and pharmacists. During outsourcing, pharmacists can be shared between the headquarters and the logistics provider.

④ Contributing to Corporate Social Responsibility (CSR)
The implementation of an inventory management system enables reliable and rapid product tracing, contributing to the fulfillment of corporate social responsibility.

⑤ Reduced Costs via On-site Constant-Temperature Storage
By storing temperature-sensitive products in constant-temperature storage within our premises, we reduced costs (freight, handling, etc.) previously incurred for transfers to specialized warehouses.
 Service Q&A
Service Q&A

We have organized frequently asked questions from the pharmaceutical industry and our responses regarding our 3PL services.
Sakata Warehouse offers one-stop solutions, ranging from business analysis, problem identification, and improvement proposals via logistics improvement consulting (paid) to actual business execution (Warehousing.3PL Services). Please feel free to consult us on any logistics matters, such as logistics improvement, logistics system restructuring, and cost control
In addition to inventory.location management, we manage manufacturing lot.receiving date.expiration date management essential for pharmaceuticals/medical devices. Based on this information, we strictly implement FIFO (First-In First-Out) practices.
In addition to barcode inspection of incoming products, we verify and register product lots upon receipt and manage storage shelf locations by product lot. We also support the sorting and shipping of non-stock back-ordered items to temporary locations, as well as reconciliation with arrival schedules/purchase data for items without barcodes.
Logistics for pharmaceuticals/medical devices involves human lives, demanding extremely high precision compared to other industries. Some products may be delivered by salespeople and require witnessed inspections. At our logistics centers, we centrally manage picking and packing by salesperson/sales office and provide delivery services to your sales offices. Please feel free to consult us.
We handle visual inspections, domestic manufacturing label (responsibility label) attachment, product storage after shipping judgment, and manufacturing record management for imported medical devices/drugs. Managed within our manufacturing (Packaging,Labeling,Storage) licensed warehouses and under our responsible technical supervisors, you can leave manufacturing tasks and domestic distribution center operations to us as a one-stop service.
Using our WMS, we perform volume calculations for loose shipping packing, pre-determine package counts, issue SCM labels, and create ASN (Advanced Shipping Notice) information. We also issue various special-purpose vouchers vouchers and manage lot performance and history records for OTC package shipping by delivery destination.
 Overview.3PL Service List
Overview.3PL Service List

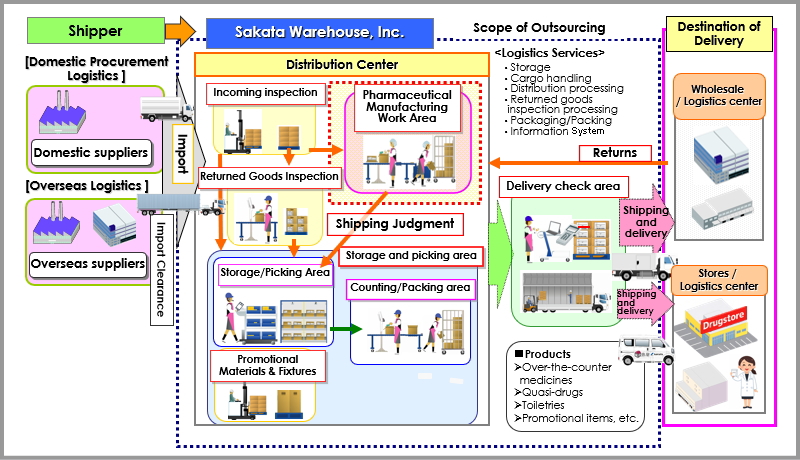
Sakata Warehouse proposes and supports the realization of integrated Supply Chain Logistics from domestic suppliers to end users. We introduce our 3PL services for the pharmaceutical industry.
Overall Image of Logistics Outsourcing
(1) Warehousing.3PL Services

① Inventory Management (Storage)
Inventory is managed by our independently developed WMS, which has a proven track record in pharmaceutical logistics. Integrated with receiving.shipping and location management, it enables real-time physical management at the SKU (piece) level. We also ensure strict manufacturing lot and temperature management required for pharmaceuticals.

② Transport and Delivery
We select and combine optimal transport modes using simulation systems. Through EDI tracking with multiple carriers, we respond quickly to retail inquiries. We also provide delivery info via email for mass shipments like new product launches. Low-temperature and constant-temperature transport are available through multiple alliance partners.

③ Piece Picking
While pharmaceutical distribution is centralizing for large wholesalers, OTC medicines are seeing a trend toward smaller lots for direct retail delivery. The Sakata Group selects material handling equipment tailored to customer needs to standardize tasks and improve accuracy. Our labeling system also integrates manual picking of irregular or long promotional items into the system.

④ Value-Added Services
Our skilled operators handle detailed value-added tasks, including barcode label printing.application, pricing, and shrink wrapping to meet various delivery requirements. We can also handle the sub-packaging of products per delivery destination as needed.

⑤ Information Systems
In addition to WMS, we manage total operations via ordering systems, labeling systems linked to material handling equipment, inspection systems for each shipping step, and transport management systems to deliver products to end users.

⑥ Quality Management
We provide continuous logistics quality and services through daily quality management, setting quality goals, and creating/executing improvement plans via PDCA.
(2) PMD Act Compliance.Manufacturing

Manufacturing (Packaging.Labeling.Storage)
At our licensed warehouses, we perform tasks based on the PMD Act, including inspections of imported cosmetics and quasi-drugs, repackaging, labeling, and manufacturing record creation by technical supervisors.
 3PL IT System Overview
3PL IT System Overview

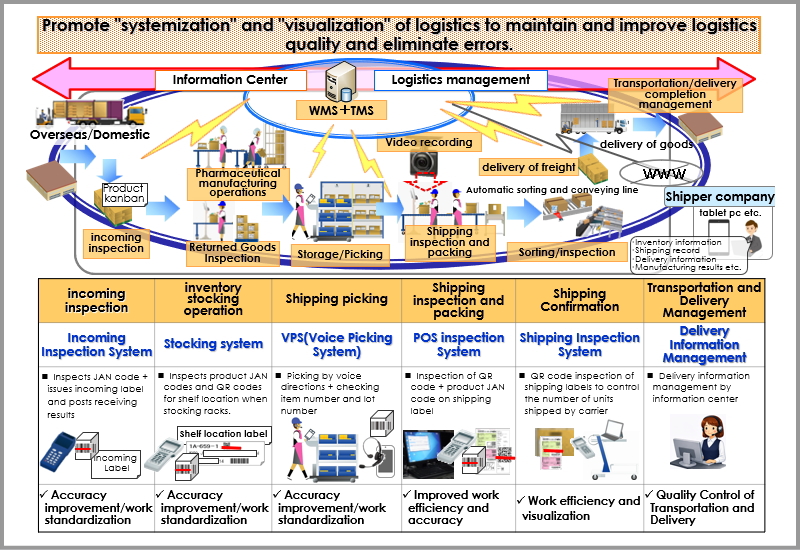
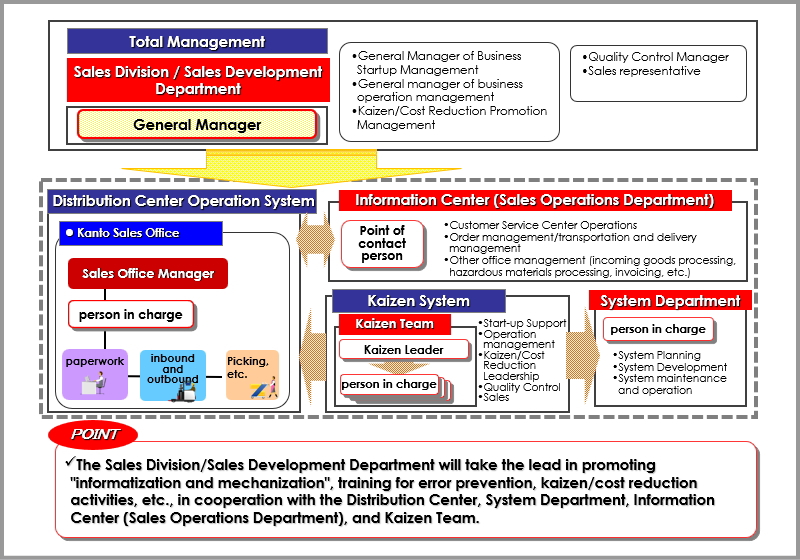
Sakata Warehouse promotes systematization and visualization of logistics to maintain quality and achieve zero errors. We link receiving.shipping, inventory, and delivery management for OTC medicines, quasi-drugs, and toiletries through core systems like WMS/TMS and warehouse support tools. We also provide high-quality information services (Fulfillment Services) through web-based ordering and shipping history tools.
3PL Logistics System Overview (Standardization.Efficiency.Visualization)
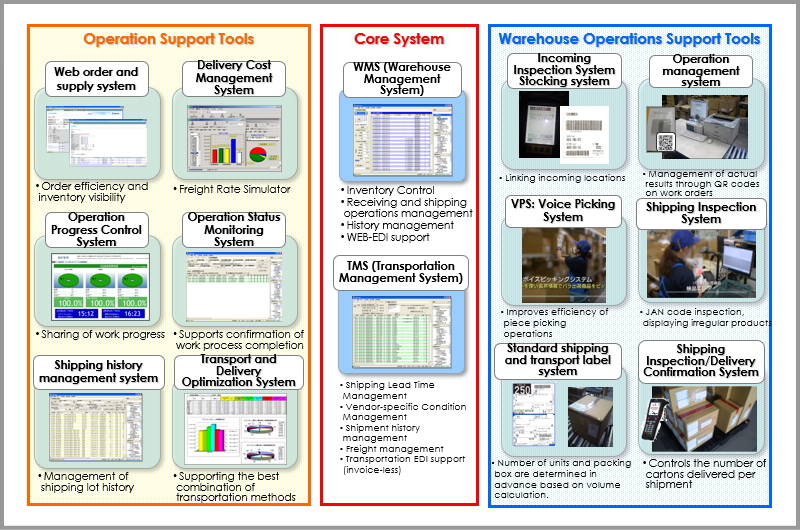
Major System Introduction
 List of Locations
List of Locations

Sakata Warehouse operates centers in Kansai, Chubu, and Kanto, providing warehouse functions that comply with the PMD Act for cosmetics, quasi-drugs, and medical devices. We also provide BCP-compliant logistics services through the networking of each location.
 Quality Management Overview
Quality Management Overview

Sakata Warehouse has established a department for company-wide quality management. We strive to improve total logistics quality through education, guidance, and support for 5S and Kaizen activities at each center. We also hold regular meetings with customers to analyze causes of claims and report on Kaizen activities to ensure stable quality management.
 Value.Strengths
Value.Strengths Service Q&A
Service Q&A Overview.3PL Service List
Overview.3PL Service List 3PL IT System Overview
3PL IT System Overview List of Locations
List of Locations Quality Management Overview
Quality Management Overview